Abstract
Introduction
Brexucabtagene autoleucel (brexu-cel) is a second generation, CD19-targeted Chimeric Antigen Receptor (CAR) T-cell therapy approved for relapsed or refractory (R/R) mantle cell lymphoma (MCL). The pivotal phase 2 trial ZUMA-2 enrolled 74 patients and infused 68, with an overall response rate (ORR) of 85% (complete response rate, 59%) among all patients who underwent apheresis (intention-to-treat, ITT). Grade 3 or higher cytokine release syndrome (CRS) and neurologic events occurred in 15% and 31% of patients, respectively. However, very little is known regarding its safety and efficacy in the real-world setting. In our study, we report clinical outcomes of patients with R/R MCL treated with commercial brexu-cel.
Methods
Data were collected retrospectively from all consecutive patients with R/R MCL who underwent apheresis for brexu-cel at 7 European sites, in 3 different countries, from start of the Compassionate Use Program in Europe (February 2020) until June 2021. Evaluable patients included those who received a CAR-T infusion and had at least 1 month of follow-up. Adverse events after infusion were graded according to the ASTCT consensus and efficacy outcomes were assessed according to Lugano criteria. Efficacy outcomes were calculated in the patients who received a CAR T-cell infusion and in all patients who underwent apheresis for brexu-cel (ITT).
Results
During the study period 28 patients with R/R MCL underwent apheresis for brexu-cel. At data cutoff, 19 (68%) patients had received a CAR T-cell infusion whereas 9 (32%) had not due to progressive disease (n=4), pending manufacturing process (n=4) or achieving a complete response with bridging (n=1). Baseline characteristics of the whole cohort and the infused patients are summarized in Table 1. Among infused patients, median age was 67 years (range 51-78) and 89% were male. Most of the patients had a high-risk simplified Mantle Cell Lymphoma International Prognostic Index score (63%), an advanced stage (84%) and 32% had received a prior autologous stem cell transplant. Median follow-up after CAR T-cell infusion was 5 months (range 1-10).
Fifteen patients (79%) received bridging therapy after apheresis, including ibrutinib in 8/15 (53%) patients, immunochemotherapy in 7/15 (47%) and radiotherapy in 6/15 (40%). Half the patients (53%) had progressive disease as best response to bridging; 4 (27%) patients had stable disease, 2 (13%) partial response and 1 (7%) achieved a complete response. Median time from apheresis to brexu-cel delivery was 30 days (range 22-48) and median time from apheresis to infusion was 42 days (range 28-77).
Among the infused patients, 17 (89%) and 12 (63%) developed any grade of CRS and neurotoxicity, respectively. Grade >2 CRS and neurotoxicity events occurred in 1 (5%) and 5 (26%) patients, respectively. Tocilizumab was administered to 16 (84%) patients and steroids to 12 (63%) patients. Two (11%) patients required admission to the Intensive Care Unit for grade 4 neurotoxicity and septic shock, respectively. At data cutoff, all infused patients were alive except for 1 patient who died due to progressive disease. Other adverse events are summarized in table 2.
Best response achieved among the infused patients included complete remission in 13 (68%) patients and partial remission in 4 (21%) patients, with an ORR of 89%. Stable disease and progressive disease were the best response in 1 (5%) patient each.
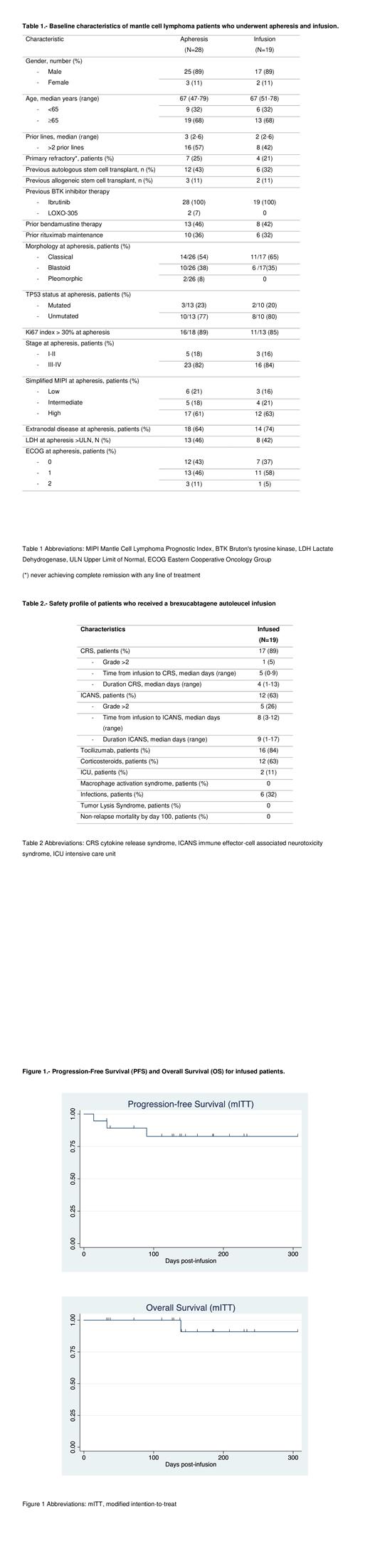
Median progression-free survival (PFS) and overall survival (OS) for infused patients were not reached with the current follow-up (Figure 1); estimated 6-month OS was 91% (95%CI 50.8-98.6) and 6-month PFS was 83% (95%CI 55.4-94.2). Three (16%) patients progressed at days 14, 33 and 90 post-infusion. In the univariate analysis, the percentage of patients with complete response was consistent across key subgroups with high-risk features (including blastoid morphology and TP53 mutation). Response rate and survival analysis by ITT will be presented at the meeting when all products still undergoing manufacturing are complete.
Conclusion
This multicenter, international study confirms that treatment with brexu-cel in patients with R/R MCL in the real-world setting has very promising efficacy, including in high-risk patients. The early safety profile was manageable and similar to the pivotal trial. Longer follow-up is needed to better assess long-term efficacy and ITT analysis.
Iacoboni: BMS/Celgene, Gilead, Novartis, Janssen, Roche: Honoraria. Chiappella: Takeda: Other: advisory board; Clinigen: Other: lecture fee, advisory board; Novartis: Other: lecture fee; Gilead Sciences: Other: lecture fee, advisory board; Janssen: Other: lecture fee, advisory board; Roche: Other: lecture fee, advisory board; Servier: Other: lecture fee; Celgene Bristol Myers Squibb: Other: lecture fee, advisory board; Astrazeneca: Other: lecture fee; Incyte: Other: lecture fee. Corral: Gileqd: Honoraria; Gilead: Consultancy; Novartis: Consultancy. Bastos-Oreiro: BMS-Celgene: Honoraria, Speakers Bureau; Gilead: Honoraria; Janssen: Honoraria, Speakers Bureau; F. Hoffmann-La Roche: Honoraria, Research Funding, Speakers Bureau; Takeda: Speakers Bureau; Novartis: Honoraria, Speakers Bureau; Kite: Speakers Bureau. Schmidt: Novartis: Consultancy, Honoraria, Other: Travel, Accommodations, Expenses; Kite/Gilead: Consultancy, Honoraria, Other: Travel, Accommodations, Expenses, Research Funding; Takeda: Consultancy, Other: Travel, Accommodations, Expenses; BMS: Consultancy, Other: Travel, Accommodations, Expenses; Bayer Healthcare: Research Funding; Janssen: Other: Travel, Accommodations, Expenses. Carpio: Regeneron, TAKEDA, Celgene, Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travels and accommodation. Reguera: Janssen, Kite/Gilead, Novartis: Speakers Bureau; BMS-Celgene, Novartis: Membership on an entity's Board of Directors or advisory committees. Kwon: Gilead: Honoraria. Martín García-Sancho: Janssen: Honoraria, Research Funding; Novartis: Consultancy; Gilead: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Roche: Consultancy, Honoraria. Zinzani: Beigene: Other, Speakers Bureau; MSD: Consultancy, Other: Advisory board, Speakers Bureau; TG Therapeutics: Other: Advisory board, Speakers Bureau; TAKEDA: Other: Advisory board, Speakers Bureau; ROCHE: Other, Speakers Bureau; EUSAPHARMA: Consultancy, Other, Speakers Bureau; SANDOZ: Other: Advisory board; SERVIER: Other: Advisory board, Speakers Bureau; BMS: Other: Advisory board, Speakers Bureau; JANSSEN-CILAG: Other: Advisory board, Speakers Bureau; GILEAD: Other: Advisory board, Speakers Bureau; CELLTRION: Other: Advisory board, Speakers Bureau; KYOWA KIRIN: Other, Speakers Bureau; NOVARTIS: Consultancy, Other, Speakers Bureau; ADC Therap.: Other; Incyte: Other, Speakers Bureau; VERASTEM: Consultancy, Other: Advisory board, Speakers Bureau. Subklewe: Pfizer: Consultancy, Speakers Bureau; Janssen: Consultancy; Takeda: Speakers Bureau; Klinikum der Universität München: Current Employment; Gilead: Consultancy, Research Funding, Speakers Bureau; Miltenyi: Research Funding; MorphoSys: Research Funding; Novartis: Consultancy, Research Funding, Speakers Bureau; Roche: Research Funding; Seattle Genetics: Consultancy, Research Funding; BMS/Celgene: Consultancy, Research Funding, Speakers Bureau; Amgen: Consultancy, Research Funding, Speakers Bureau. Barba: Novartis: Honoraria; Pfizer: Honoraria; Gilead: Honoraria; BMS: Honoraria; Amgen: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal